Von Gierke disease stands for a large group of glycogen storage disorders (GSDs). Generally, storage diseases are a class of medical conditions in which stores glycogen cannot be metabolized into glucose in order to supply energy and maintain a constant blood glucose level for the body.
Von Gierke disease is presently referred to as Type 1 glycogen storage disease (GSD I). It is a genetic condition that is inherited as an autosomal recessive disorder, that is, it occurs when a person inherits a non-working gene from each parent.
If a person receives one working gene and one non-working gene for the disease, the person will be a carrier for the disease but will not show any symptoms.
The probability for two carrier parents to both pass the defective gene and therefore have a child with von Gierke’s disease is 25% with each pregnancy.
The probability to birth a child who is a carrier just like the parents is 50% with each pregnancy while the chance for a child to receive working genes from both parents is 25%. This probability is the same for both males and females.
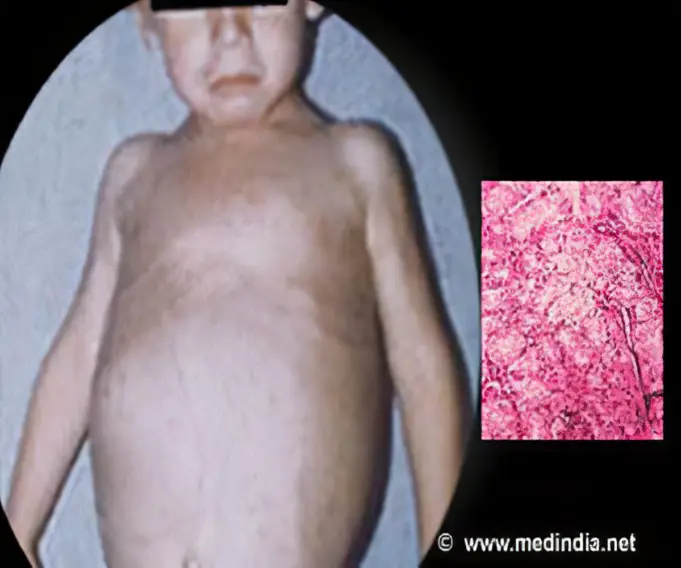
Von Gierke disease is characterized by accumulation of excessive glycogen and fat in the liver and kidneys that may result in the enlargement of the organs and growth retardation leading to a short stature.
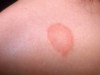
Hypoglycemia is a major feature in the case of Von Gierke disease and abnormalities of lipids may lead to xanthoma formation (a small, yellow nodule, rich in cholesterol and other lipids, that occurs in the skin often close to the joint).
Von Gierke disease was first described by Von Gierke in 1929 in a seminal article detailing his discovery of a newly described glycogen storage disease that affected the liver and kidneys that he discovered when conducting an autopsy on an affected child.
The disease was originally named “Hepato-Nephromegalia glykogenica” by Von Gierke. Von Gierke’s discovery was later rewarded and the disease was later given the eponymous distinction of being known as von Gierke disease, which was subsequently renamed to glycogen storage disease type 1.
He described the related medical disorder as an enzyme defect in glucose-6-phosphatase so that glucose-6-phosphate cannot be converted into free glucose but is metabolized to lactic acid or incorporated into glycogen.
Presentation of the condition is usually immediately after birth but in few cases, the disease has been observed to present itself a little later. Shortly after birth, hypoglycemia and lactic acidosis often cause convulsions, irritability, pallor, cyanosis, hypotonia and apnoea.
Some children may experience diarrhea due to pseudocolitis and a characteristic rounded ‘doll’s face’ due to fat deposition. During the first weeks of life, the liver is normal in size, but it enlarges, sometimes very considerably to cause marked abdominal distention.
Growth is also retarded, and height is usually below the third centile, also puberty is delayed but the mental development of the child remains normal.
Von Gierke Disease Classification
Von Gierke’s disease is universally classified into four subgroups:
- Glucose-6-phosphatase deficiency is the cause of GSD Type Ia and should not be confused with glucose-6-phosphate dehydrogenase deficiency.
- Another type of von Gierke’s disease is GSD type Ib which is as a result of a specific translocase deficiency. Persons with type Ib have altered neutrophils function predisposing them to Gram-positive bacterial infections.
- Two other translocase deficiencies have been described to give types Ic and Id.
There are however two major forms for practical purposes. Type Ia has deficient glucose-6-phosphatase in the liver and type Ib has normal activity.
Recently, the prevalence of Von Gierke disease is unknown and the incidence of GSD I (Von Gierke disease) in the overall population is 1 per 100,000 with GSD Ia and GSD Ib prevalent in 80% and 20% respectively.
The Ashkenazi Jewish population has a 5-times greater prevalence compared to rest of the population.
Causes of Von Gierke Disease
Von Gierke’s disease is inherited as an autosomal recessive genetic disorder and it is related with abnormalities in two genes.
Mutations in the G6PC gene on chromosome 17 result in a deficiency in the glucose-6-phosphatase (G6Pase) enzyme and account for 80% of glycogen storage disease type I; this type of von Gierke’s disease is termed GSD type Ia.
Mutatuins in the SLC37A4 gene result in a deficiency in the glucose-6-phosphatase translocase enzyme (transporter deficiency) and it accounts for 20% of GSDI, it is also known as GSD type Ib.
Both of these enzyme deficiencies cause excess amounts of glycogen along with fats to be stored in the body tisssues.
Symptoms of Von Gierke Disease
Investigations from early research into Von Gierke’s disease identified so many clinical manifestations falsely thought to be primary features of the genetic disorder.
But subsequent research revealed that these clinical features are the consequences of only one or two fundamental abnormalities:
- Inability of the liver to covert stored glycogen into glucose through glycogenesis.
- In GSD type Ib, impairment of the neutrophils’ ability to take up glucose, leading to neutrophils dysfunction and neutropenia (low neutrophil count).
The above abnormalities result to a small number of primary clinical manifestations, which are the features considered in diagnosis of GSD type I:
- Hypoglycemia (low blood sugar), due to impairment of glycogen breakdown causing insufficient fasting blood glucose.
- Hepatomegaly of non-alcoholic fatty liver disease, due to impairment of glucogenolysis causing glycogen accumulation in the liver.
- In GSD Ib, increased infection risk, due to neutropenia and neutrophil dysfunction.
People affected with the condition commonly present with the following manifestations, eother linked to one or more of the primary clinical manifestations:
- Lactic acidosis, due to high level of lactic acid in the blood, caused by prolonged hypoglycemia.
- High levels of uric acid in the blood, which makes the individual prone to gout or kidney damage, caused by low serum insulin levels in prolonged hypoglycemia.
- Hepatic adenomas developing later in life and attendant risk to anemia, believed to be caused by blood glucose regulation in the presence of non-alcoholic fatty liver disease.
- Inflammatory bowel disease (GSD Ib) caused by neutrophil dysfunction.
Some other clinical manifestations may arise due treatment attempts of the primary clinical manifestation, they include:
- Pancreatic hypertrophy, due to increased carbohydrate intake causing frequent engagement of the insulin response.
- Splenomegaly (GSD Ib), due to the long-term use of filgrastim to treat neutropenia causing sequestration of blood factors in the spleen.
- Anemia (GSD Ib) may occur due to long-term use of filgrastim resulting in sequestration of hemoglobin in the spleen.
- The abnormal low number of platelets in the blood (GSD Ib) may occur, due to long-term use of filgrastim causing sequestration of platelets in the spleen.
Diagnosis of Von Gierke Disease
Von Gierke’s disease is medically diagnosed by laboratory blood tests that show abnormal levels of glucose, lactate, uric acid, triglycerides and cholesterol.
A critical blood specimen obtained at the time of hypoglycemia usually reveals a mild metabolic acidosis, high free fatty acids and beta-hydroxybutyrate, very low insulin levels, and high levels of glucagon, cortisol, and growth hormone.
Molecular genetic testing for the G6PC and SLC37A4 genes is also available to confirm a diagnosis. This molecular genetic testing can also be used for carrier testing and prenatal diagnosis.
Liver biopsy can be used to confirm diagnosis with an electron microscope and assay of glucose-6-phosphatase activity in the tissue for GSD Ia.
Differential Diagnosis
Differentiation of GSD I (von Gierke’s disease) from other diseases that may present similar symptoms such as hepatomegaly and/or hypoglycemia are very necessary.
Some of these clinical conditions include:
- GSD 0 (glycogen synthase deficiency
- GSD III (glycogen debranching enzyme deficiency), Forbes disease
- GSD IV (branching enzyme deficiency), Aderden disease
- GSD VI (hepatic phosphorylase deficiency), Hers disease
- GSD IX (hepatic form of phosphorylase kinase deficiency)
- GSD XI (Franconi-Bickel syndrome due to glucose transporter protein II deficiency)
- Hepatitis (primary liver disease)
- Niemann-Pick B disease
- Hereditary fructose intolerance
- Gaucher disease
- Disorders of Gluconeogenesis (Frutose-1,6-bisphosphatase deficiency)
Complications
- Patients with von Gierke’s disease may develop disorders from impaired platelet function.
- There is an increased risk of osteoporosis and fracture due to Vitamin D deficiency
- Renal failure may occur due to proximal renal tubular or renal glomerular dysfunction.
- Development of anemia of chronic kidney disease due to prolonged renal failure, that may be exacerbated by iron deficiency.
- Increased risk of Crohn’s disease-like enterocolitis and hypothyroidism.
- Menorrhagia (excessive menstruation) and polycystic ovaries in female and gout from hyperuricemia
- Severe hyperlipidemia and hypertriglyceridemia increase the risk of xanthoma formation, acute pancreatitis and early atherosclerosis.
- Hepatocellular carcinoma
- Short Stature
Treatment/Management of Von Gierke Disease
The main aim for the management and treatment of von Gierke’s disease are the prevention of acute metabolic derangement, prevention of acute and long-term complications, attainment of normal psychological development and good quality lifestyle.
Drugs and Surgery
- Blood loss may require oral iron.
- Raised uric acid levels may require allopurinol. Treatment of hyperuricemia and pyelonephritis protects renal function.
- Diazoxide to maintain blood glucose has been disappointing.
- Liver transplantation improves metabolic control and seems effective, although the immunosuppression may cause deterioration of renal function.
Diet and Lifestyle
- Young infants require continuous nasogastric tube feeding.
- Older infants and children are recommended to include corn-flour in their diet to ensure slow release of glucose by day but nasogastric feeding by night is still often required to prevent hypoglycemia and associated metabolic problems.
- Restriction of lipids in advised but statins are not used.
- Intake of fructose and galactose should be restricted, as they do not increase glucose levels but increase lactic acid production.
- Physical activities and exercises do not have to stop but rough games, contact sports and strenuous exercises should be avoided because of bleeding tendencies and risk of rupture to an enlarged liver.
Summary
Individuals with von Gierke’s disease should always be monitored at least annually with kidney and liver ultrasound and routine blood work specifically used for monitoring GSD patients.
Also seeking Genetic counseling is not a bad idea, it is actually recommended for affected individuals and their families. Early diagnosis and effective treatment programs have proven to work wonders; resulting in normal growth and puberty and many affected persons live into adulthood and enjoy normal life activities.
Many female patients have also had successful pregnancies and child delivery.
Sources;
- Von Gierke Disease; https://medlineplus.gov/ency/article/000338.htm
- Von Gierke Disease; https://www.mountsinai.org/health-library/diseases-conditions/von-gierke-disease
- Von Gierke Disease; https://www.britannica.com/science/von-Gierkes-disease
- Von Gierke Disease; https://liverfoundation.org/for-patients/about-the-liver/diseases-of-the-liver/type-i-glycogen-storage-disease/