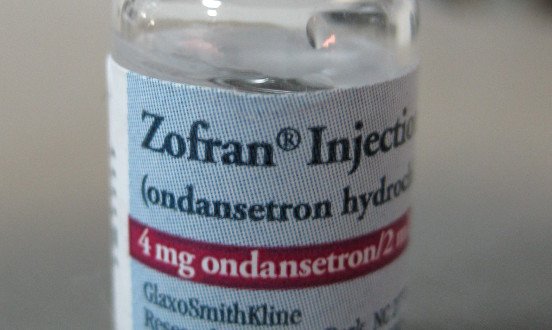
Zofran, also known generically as ondansetron, is a drug that was developed in the mid 1980s by GlaxoSmithKline (GSK). The chemical ondansetron hydrochloride (C18H19N3O) belongs to a class of medications known as Serotonin-3 (5-HT3) Receptor Antagonists and was developed in London around 1984. [1] Zofran later received a United States patent in 1991, which was enforceable until December 2006. By 2006, Zofran had become the 20th highest selling drug in the United States.
After the patent expired, the generic version of ondansetron became available to produce in the United States, and the pharmaceutical companies Teva and SICOR were the first ones to be granted FDA approval to market and sell their generic forms of Zofran.
How Does it Work?
Zofran works by reducing the activity of the “vomiting center” in the brain, which is activated by the vagus nerve. Zofran targets the vagus nerve and blocks the receptors that receive serotonin, a substance that can trigger vomiting or nausea. Essentially, if the nausea receptors are blocked, it cannot send signals to the brain telling the body to feel nauseous.
Zofran and the FDA
The FDA approved GSK’s Zofran to treat nausea and vomiting in surgery and cancer patients only. However, Zofran has also been used “off-label” to treat morning sickness in pregnant women. Although Zofran has not yet been FDA approved for treatment in pregnant women, it is frequently prescribed to pregnant women to treat nausea.
Prior to Zofran, Bendectin was an FDA approved drug that was used from 1956 – 1983 to treat pregnant women suffering from morning sickness. In 1983, the makers of Bendectin removed it from the market after the company began facing numerous lawsuits due to a possible link to birth defects, though this link was generally disproven. [2] Bendectin later returned to the market under a new trademark name, Diclegis, and became FDA approved in 2013. The lack of an FDA approved drug to treat morning sickness between 1983 and 2013 encouraged doctors to commonly prescribe drugs, like Zofran, off-label to reduce nausea in pregnant women.
The FDA has supplied multiple warnings about the potential link between Zofran and babies born with birth defects. In 2013, the FDA declared that the effects of Zofran and its safety for use during pregnancy are unclear, and that the use of the drug has been linked to Serotonin Syndrome. [3] Data from the National Birth Defects Prevention Study revealed the possible connection between ondansetron and babies born with an isolated cleft palate, among other birth defects. [4]
Birth Defects Associated with Zofran
Several birth defects found to be associated with Zofran use include:
- Cleft Lip and Cleft Palate
- Heart malformations
- Musculoskeletal Anomalies and Disorders
- Developmental Delays
- Jaundice
- Miscarriages
Impact of Zofran Birth Defects
Cleft Lip and Palate Impact:
Cleft lip and cleft palate occur when there is a lack of tissue in the mouth causing an improper formation between the lip and mouth. This can result in a partial or entire split in the roof of the mouth. Babies born with these types of oral cleft defects will often require corrective surgery.
Additionally, both cleft lip and palate can make it difficult for a child to eat and talk. Children with oral clef defects are especially susceptible to middle ear infections, which can also lead to hearing loss.
Heart Defect Impact:
AVSD are a family of congenial heart defects. An atrioventricular septal defect can cause the failure of formation of certain structures in the heart known as the atrial septum and ventricular septum. This failure of formation results in “holes” in the atrial and ventricular septa leading to an increased workload on the heart. This makes breathing more difficult and causes the infant to tire while feeding.
Other potential impacts of AVSD include slower growth, decreased energy, and the inability to gain weight. Symptoms of ASVD usually appear between 4 to 8 weeks of age, and are often identified initially by a heart murmur.
Summary
Zofran is a prescription medication that has been prescribed off-label by the pharmaceutical drug manufacturer, GlaxoSmithKline. It has been linked to various birth defects in children.
For more information on Zofran and the potential birth defects related to its use, please visit our Zofran and birth defects page.
Author bio:
My name is Charles Brown and I have the website, www.birthinjuryguide.org. If you need information about birth defects in children that have resulted from Zofran use during pregnancy, visit our website today for additional information.