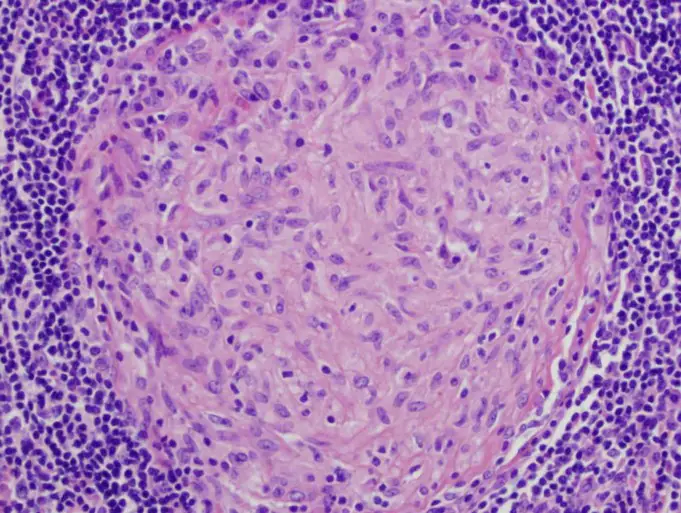
Granuloma is a term for a small area of inflammation; it is typically a benign (noncancerous) type of defensive reaction that triggers the body to attack foreign invaders such as bacteria and other foreign bodies.
The granulomatous disease is also referred to as chronic granulomatous disease which is primarily a condition that entails deficiencies in the immune system of the body.
Chronic granulomatous disease (CGD) is a rare inherited condition due to defects in neutrophils and macrophages in the blood, leading to the inability of cells that ingest and destroys microorganisms, known as phagocytes, to secrete chemicals necessary to destroy some particular types of bacteria and fungi.
Chronic Granulomatous disease is characterized by an inability of the immune system to resist infections caused by these particular kinds of bacteria and fungi and a tendency to develop chronic inflammations.
The most common form of chronic granulomatous disease is caused by a mutation in an X-chromosome-linked gene and this account for about 80% of all cases. Granulomatous disease was first observed over two centuries ago during the autopsy of a tuberculous lung.
The first recorded medical case dates back to 1950, four boys from Minnesota were consecutively diagnosed of the condition and it was later in 1957 named “a fatal granulomatous of childhood” in a published article describing the boys’ condition.
The underlying cellular abnormalities causing the disease were revealed in 1967, and since then further conducted research has clarified the molecular mechanisms of the disease. The chronic granulomatous disease was known and labeled for these masses of inflammation before the discovery of its link to genetics.
In chronic granulomatous cases, phagocytic cells lose their ability to cooperatively produce superoxide– a compound formed when oxygen is ionized, this compound is formed by the action of an enzyme, superoxide dismutase whose action is coordinated by the X-chromosome linked protein, this enzyme constitutes a very important antioxidant defense against oxidative stress in the body.
The disease usually manifest within the first year after birth, however, early diagnosis permits intervention to improve the limited life expectancy.
Statistics shows that only 50% of children diagnosed of chronic granulomatous disease live pass 25 years. Because of the systems inability to fight off infections, children with chronic granulomatous disorder always get very sick from bacterial infections that could be mild or even harmless on healthy bodies.
These infections may affect the lungs, liver, genitourinary tract, gastrointestinal tract, and even lymph nodes, leading to the development of masses of inflammatory tissues in response to chronic infections.
Chronic granulomatous disease is a very rare condition affecting approximately 1 in 250,000 to 500,000 births, with most affected persons been males. Bacteria and fungi such as Staphylococcus aureus, Burkholderia cepacia complex, Serratia marcescens, Nocardia, candida and Aspergillus are some types of microorganisms that are not affected by the immune system.
Classification of Granulomatous Disease
Chronic granulomatous disease (CGD) is an umbrella descriptive name for a genetically heterogeneous group of immunodeficiency disorders.
The core etiology of these disorders is the failure of phagocytic cells to destroy pathogens due to a defective system of enzymes that produce free radicals and other toxic secretions.
The types of CGD include:
- X-linked CGD
- Autosomal recessive cytochrome b-negative CGD
- Autosomal recessive cytochrome b-positive CGD type I
- Autosomal recessive cytochrome b-positive CGD type II
- Atypical granulomatous disease
Causes of Granulomatous Disease
Research conducted and deductions from observed cases suggest that there are five distinct genetic mutations associated with the condition.
In 70% of cases, this mutation is X-linked and only manifest in males. The other four mutations are located separately on different chromosomes and inherited in an autosomal recessive pattern.
About 15% of these cases are females. In cases associated in mutations of the X-linked chromosomes, the cause of the disease is phagocytic cells losing their ability to produce superoxide; phagocytic cells include monocytes, macrophages, neutrophils, mast cells, and tissue dendritic cells, among these cells, cell abnormality is mostly found in macrophages and neutrophils.
The neutrophils can no longer effectively attack certain pathogens due to a defective function of the nicotinamide adenine dinucleotide phosphate (NADPH) oxidase, which produces hydrogen peroxide.
The hydrogen peroxide is then metabolized to reactive oxygen which is necessary for the phagocyte respiratory burst that kills ingested pathogens.
In few fungi and bacteria such as candida, staphylococcus and others earlier mentioned, the reduced amount of metabolized hydrogen peroxide due to the defective NADPH oxidase is ineffective and it is quickly neutralized by a pathogen-inherited catalase.
In the course of an infection, patients suffering from CGD develop granulomas as a result of a dysregulated inflammatory response potentially leading to obstructive lesions.
Symptoms of Granulomatous Disease
Typically, both patients with X-linked chromosomal mutation (signs are noticed first year of age) and those with autosomal recessive pattern (may begin during adolescence) forms of chronic granulomatous disease, will suffer reoccurring episodes of infections due to the decreased capacity of their immune system to fight off pathogens.
These acquired infections are specific and in decreasing order of frequency include:
- Pneumonia
- Abscesses of the skin, tissues and organs
- Septic arthritis
- Oseteomyelitis
- Bacteemia/fungemia
- Superficial skin infections such as cellulitis or impetigo.
- Other symptoms of CGD include:
- Enlargement of the liver and spleen
- Slow growth rate in children
- Inflammation of the intestines: this occurs in 40-50% of patients with CGD and it is sometimes mistaken for Crohn disease.
Infants with chronic granulomatous disease presents with:
- Dermatitis
- Gastrointestinal obstruction
- Colitis with resulting diarrhea
- Failure to strive.
- Recurrent infections
A small group of chronic granulomatous patients may also be affected by McLeod syndrome due to the proximity of the two genes on the same X-chromosome.
Diagnosis of Granulomatous Disease
Diagnosis for chronic granulomatous disease involves blood tests and sometimes genetic testing; blood samples are collected and sent to a laboratory.
If CGD is suspected, neutrophil function tests are carried out, the tests include; nitroblue tetrazolium (NBT) reduction test, dihydrohodamine (DHR) 123 test, direct measurement of superoxide production, cytochrome C reduction and chemiluminescence.
DHR test is always preferred because it is easy to use and it is able to distinguish between X-linked and autosomal forms of CGD. The NBT test is the original and most widely known neutrophil-function test for CGD. It is negative in the presence of CGD that is it does not turn blue.
The higher the blue gradient score, the better the cells are at producing superoxide (reactive oxygen species). The tests depend on the direct reduction of NBT to the insoluble blue compound formazan by superoxide which is produced by normal peripheral blood neutrophils stimulated in vitro.
DHR 123 test entails the stimulation of the respiratory burst of the neutrophils with phorbol myristate acetate (PMA), resulting in oxidation of dihydrorhodamine 123 (nonflourescent derivative of rhodamine) to rhodamine 123 (green, fluorescent compound), which can be measured by flow cytometry.
Abnormality of the test is an indication of CGD causes, no shift in fluorescence with stimulation. This test also quantitatively allows differentiating oxidase-positive from oxidase-negative phagocytes in subpopulations in chronic granulomatous disease patients and identifies deficiencies in gp91phox and p47phox.
Genetic testing is used to confirm chronic granulomatous disease once it is diagnosed based on abnormal neutrophil function test. Genetically, p47phox defect is usually difficult to identify because eit is caused by pseudo-gene conversion and it may be missed in typical sequencing studies. In this case, immunoblotting or flow cytometry can shoe absence of proteins.
Also, Parental testing is necessary when a family member has already been diagnosed with chronic granulomatous disease. This test analyses NADPH oxidase activity of neutrophils from fetal blood.
A sample taken from amniotic fluid or chorionic villi provides an early and more reliable diagnosis for families at risk.
Treatment of Granulomatous Disease
Treatment approaches towards chronic granulomatous disease include the following:
- Antibiotics usually sulfamethoxazole and trimethoprim, antifungal drugs such as itraconazole, are usually administered to prevent and treat bacterial and fungal infections.
- Interferon gamma is a drug used to modify the immune system, it is injected thrice a week and it helps to reduce the number and severity of infection.
- Transfusion of granulocytes (a type of white blood cell that includes some phagocytes) can be a life saver when other treatment options are ineffective.
- Stem cell transplantation: so many patients have been cured of chronic granulomatous disease with stem cells transplant. Blood relatives who have similar tissues and blood types are best donors. Before transplantation commences, the doctor administers immunosuppressants to suppress the immune system of the patient and help prevent rejection of the transplant.
Summary
There are presently no studies to reveal information on the long-term outcome of chronic granulomatous disease with modern treatment. Without prompt treatment, children often die in the first ten years of their life.
The increased severity of X-linked CGD has led to a decrease in the survival rate of patients as 20% of X-linked patients die of CGD-related causes at age 10, whereas 20% of other forms of CGD die by the age of 35.
However, a suggestion from centers specializing in the infantile care of patients with CGD like the Boston Children’s Hospital Immunology Program says the mortality rate has fallen to under 3% and 1% respectively.
The quality of life and longevity for patients with CGD has improved dramatically over the last 50 years, an average patient now survives at least 40 years.
The great majority of children with chronic granulomatous disease are expected to live well into adulthood, and many adult individuals with the disease have jobs, get married, and bear children.
However, patients of chronic granulomatous disease remain at risk for infection throughout life. They must remain cautious and take their prophylaxis and get early diagnosis and treatment for possible infections.
Hospitalization may also be recommended to locate sites and cause of infections.
Sources;
- ChronicGranulomatousDisease; http://www.frontiersin.org/articles/10.3389/fimmu.2018.01258/full
- Chronic granulomatous disease; https://rarediseases.info.nih.gov/diseases/6100/chronic-granulomatous-disease
- Unusual cause of granulomatous disease; https://bmcclinpathol.biomedcentral.com/articles/10.1186/1472-6890-7-1
- Chronic Granulomatous Disease; https://www.childrenshospital.org/conditions-and-treatments/conditions/c/chronic-granulomatous-disease-cgd